Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Jo, Akinori*; Kugara, J.*; Trobradovic, H.*; Yamabe, Kazunori*; Sugo, Takanobu; Tamada, Masao; Kume, Tamikazu
Industrial & Engineering Chemistry Research, 43(7), p.1599 - 1607, 2004/03
Times Cited Count:28 Percentile:67.22(Engineering, Chemical)Fibrous iminodiacetic acid cheating cation exchangers were derived from chloromethylstyrene radiation-grafted polyethylene-coated polypropylene filamentary fiber and its nonwoven cloth. Ligand contents and acid capacities of the resulting cation exchangers were ca. 2 mmol/g and ca. 4 mmol/g for the filamentary fiber and for the non-woven cloth, respectively. The selectivity sequence of nonwoven cloth shape for dialect metal ions is Mg(II)  Ca(II)
Ca(II)  Co(II)
Co(II)  Zn(II)
Zn(II)  Cd(II)
Cd(II)  Ni(II)
Ni(II)  Pb(II)
Pb(II)  Cu(II). Capacities in mmol/g at pH 5 were Ca(II) 0.91, Mg(II) 0.98, Cd(II) 1.5, Ni(II) 1.5, Pb(II) 1.6, Cu(II) 1.8. Column mode for filamentary fiber shape revealed that breakthrough capacities for Cu(II) (ca. 1 mmol/g) were not dependent on flow rates up to 200
Cu(II). Capacities in mmol/g at pH 5 were Ca(II) 0.91, Mg(II) 0.98, Cd(II) 1.5, Ni(II) 1.5, Pb(II) 1.6, Cu(II) 1.8. Column mode for filamentary fiber shape revealed that breakthrough capacities for Cu(II) (ca. 1 mmol/g) were not dependent on flow rates up to 200  300 h
300 h in space velocity.
in space velocity.
Tanaka, Tadao; Sakamoto, Yoshiaki; Ogawa, Hiromichi
Genshiryoku Bakkuendo Kenkyu, 9(1), p.29 - 34, 2002/09
To obtain structural information on functional groups of humic substances complexing with metal ions, infrared (IR) spectral analysis of the humic substances dissolving in aqueous solutions has been studied by ATR (Attenuated Total Reflection) method. By using the ATR method, IR spectral shifts of humic substance due to complexation with metal ions were observed. The observation provided with information on complexation kinetics, and on characteristics of functional groups depending on solvent properties, such as pH and solute concentrations. These information was difficult to be obtained by conventional FTIR spectroscopy using powdered humic substances. The obtained results show that IR spectral shifts on the functional groups of the humic substances complexed with metal ions could be possibly characterized  by using the ATR-FTIR spectroscopy.
by using the ATR-FTIR spectroscopy.
Tachimori, Shoichi; Yaita, Tsuyoshi; Suzuki, Shinichi
Proc. of Workshop on Long-Lived Radionuclide Chemistry in Nuclear Waste Treatment, p.179 - 188, 1997/00
no abstracts in English
Tanaka, Tadao; Nagao, Seiya; Sakamoto, Yoshiaki; Onuki, Toshihiko; S.Ni*;
Hoshasei Haikibutsu Kenkyu, 3(1), p.41 - 47, 1996/08
no abstracts in English
 Co and
Co and  Am
AmTanaka, Tadao;
Radioisotopes, 44(2), p.99 - 102, 1995/02
no abstracts in English
Yoshida, Masaru; Asano, Masaharu; *; Kumakura, Minoru
Polymer, 31, p.371 - 378, 1990/02
Times Cited Count:17 Percentile:63.82(Polymer Science)no abstracts in English
Yoshida, Masaru; *; Asano, Masaharu; Kumakura, Minoru
European Polymer Journal, 26(2), p.121 - 125, 1990/00
Times Cited Count:12 Percentile:53.9(Polymer Science)no abstracts in English
Yoshida, Masaru; *; Asano, Masaharu; Kumakura, Minoru
Colloid and Polymer Science, 267, p.986 - 991, 1989/00
Times Cited Count:12 Percentile:56.42(Chemistry, Physical)no abstracts in English
Yoshida, Masaru; Asano, Masaharu; *; R.Chosdu*; Kumakura, Minoru
J. Polym. Sci., Part C, 27, p.437 - 442, 1989/00
no abstracts in English
Radiation Physics and Chemistry, 29(N0.6), p.469 - 475, 1987/06
no abstracts in English
 ,
, ,
, -trifluoroacrylate with tetrafluoroethylene and
-trifluoroacrylate with tetrafluoroethylene and  -olefin
-olefin; ; *; Machi, Sueo
Journal of Polymer Science; Polymer Chemistry Edition, 18, p.1325 - 1337, 1980/00
no abstracts in English
Sasaki, Yuji; Sugo, Yumi; Morita, Keisuke
no journal, ,
The effect of the alkyl substituents on amidic N atoms in diglycolamide (DGA) compounds on solvent extraction has been investigated. The purpose of this study is to examine the effect of alkyl substituents on Ln and An extraction. Diglycolamides obtained are O(CH CONR
CONR )
) with R = CH
with R = CH , C
, C H
H , C
, C H
H , C
, C H
H , C
, C H
H , C
, C H
H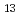 , C
, C H
H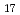 , C
, C H
H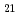 , C
, C H
H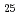 and alkyl and phenyl-groups. From the present work, it was clear that DGA with shorter alkyl chain have high solubility in water, and those with longer alkyl chain have high solubility in n-dodecane. Although DGA with short alkyl chain have high distribution ratio, DGA with long alkyl chain have high loading capacity of Nd. Even DGA derivatives, DGA with phenyl groups have not only low solubility in dodecane but also low D values for lanthanides.
and alkyl and phenyl-groups. From the present work, it was clear that DGA with shorter alkyl chain have high solubility in water, and those with longer alkyl chain have high solubility in n-dodecane. Although DGA with short alkyl chain have high distribution ratio, DGA with long alkyl chain have high loading capacity of Nd. Even DGA derivatives, DGA with phenyl groups have not only low solubility in dodecane but also low D values for lanthanides.